
Find the ratio of molar amounts of H2s needed to precipitise metal ions from. 20 ml each of 1 M CD(NO3)2 and 0.5 M.CuSO4? - EduRev Class 11 Question

KNR Copper Sulphate Powder - 900 gm Pure Neela thotha, Cuso4 Powder : Amazon.in: Industrial & Scientific

2022 Hot Sell China 99% CuSo4. H2O CuSo4.5H2O Copper Sulphate - China Industrial Grade Copper Sulphate Pentahydrate, Copper Sulphate Pentahydrate CAS 7758998 | Made-in-China.com

China Copper Sulfate Monohydrate and Pentahydrate Blue Powder Blue Copperas CuSO4 Animal Feed Additive Manufacturer and Exporter | Sustar
Find the ratio of molar accounts of H2SO4 needed to preciptise metal ions from 20ml each of 1M cd(NO3)2 and 0.5 M cuso4??

China Copper Sulfate Monohydrate and Pentahydrate Blue Powder Blue Copperas CuSO4 Animal Feed Additive Manufacturer and Exporter | Sustar

SOLVED: Write the balanced molecular chemical equation for the reaction in aqueous solution for copper(II) sulfate and ammonium hydroxide. If no reaction occurs, simply write only NR: CuSO4(aq) + 2NH4OH(aq) -> Cu(OH)2(s) + (
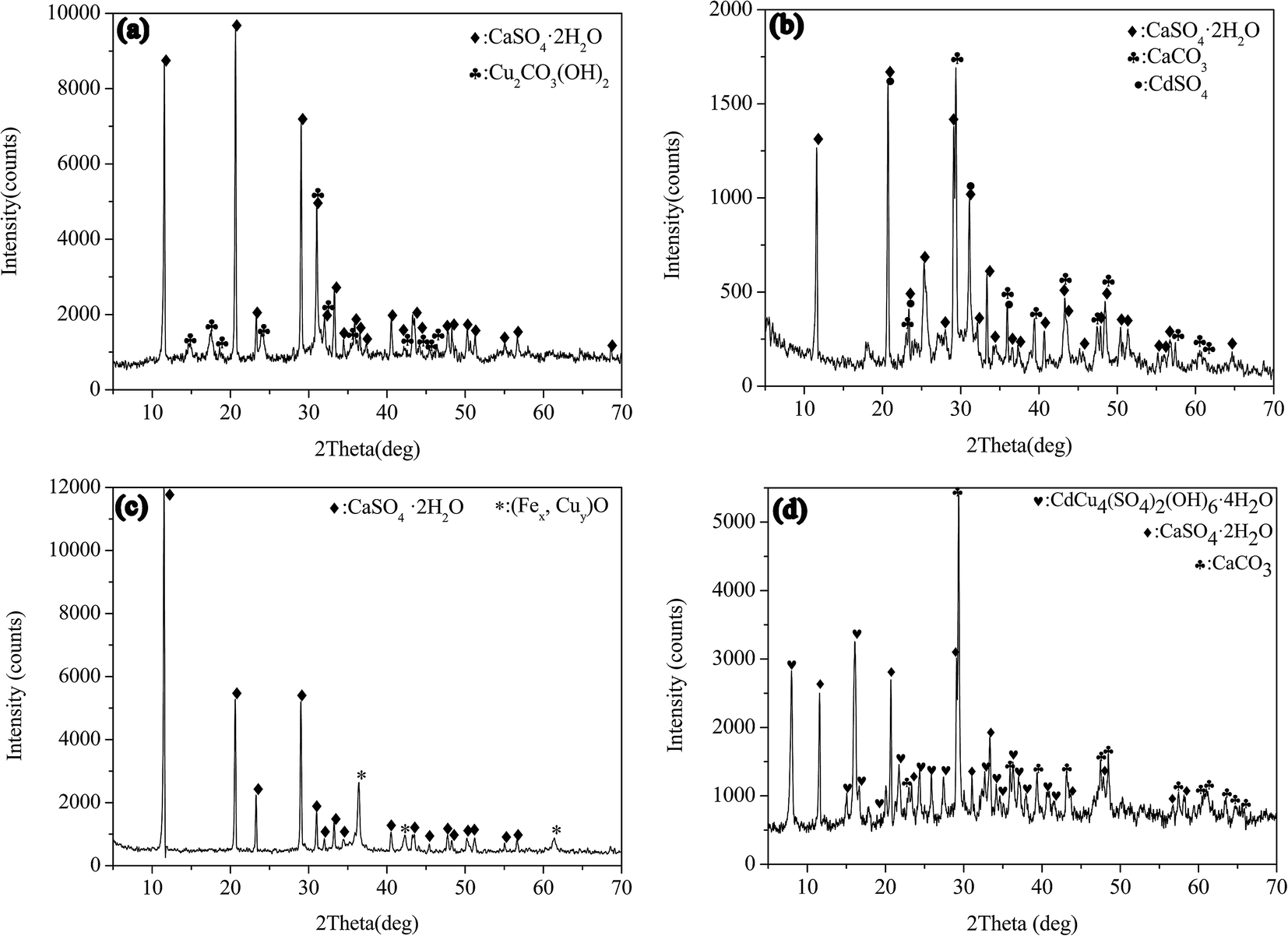
Separation of Cu( ii ) from Cd( ii ) in sulfate solution using CaCO 3 and FeSO 4 based on mechanochemical activation - RSC Advances (RSC Publishing) DOI:10.1039/C6RA25412H

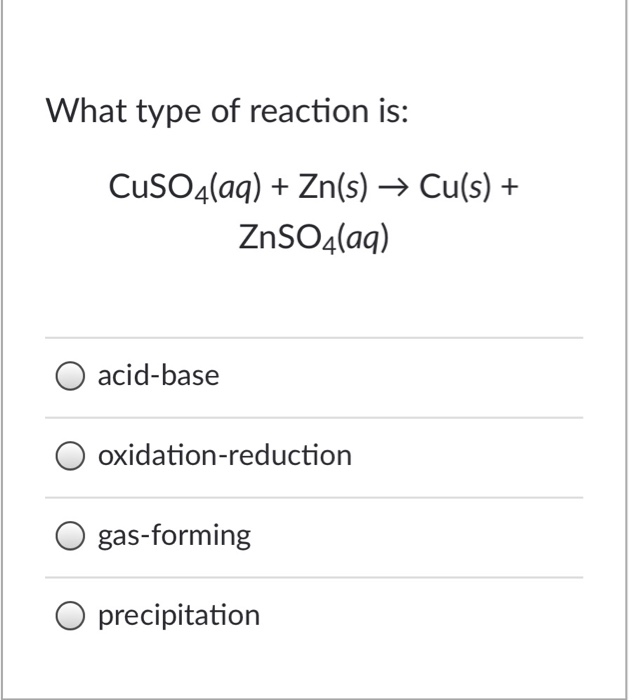
SOLVED: In which reaction is precipitation occurring? A. MgCl2(aq) + CuSO4(aq) â†' CuCl2(aq) + MgSO4(aq) B. CdSO4(aq) + K2S(aq) â†' CdS(s) + K2SO4(aq) C. NaOH(aq) + NH4Cl(aq) â†' NaCl(aq) + NH4OH(aq) D.

Find the ratioof molar amount of H2S needed to precipitise metal ions from 20ml each of 1M Cd(NO3)2 and 0.5M CuSO4 - kqs9srzz

Circular dichroism (CD) spectra of Ang60-68 + CuSO4 (black line) and... | Download Scientific Diagram







![Copper Sulfate Pentahydrate [CuSO4.5H2O] [CAS_7758-99-8] Fine Crystals – Wintersun Chemical Copper Sulfate Pentahydrate [CuSO4.5H2O] [CAS_7758-99-8] Fine Crystals – Wintersun Chemical](https://www.wintersunchemical.com/cdn/shop/products/03-030-2_1024x1024.jpg?v=1595287760)



